Urgent Care Evaluation
History and vital signs will usually identify the 50% of patients whose dizziness is caused by some general medical cause. The descriptive word used by the patient (e.g., “lightheadedness” or “vertigo” or “imbalance”) for their dizziness is not useful diagnostically and should not, by itself, drive the workup.
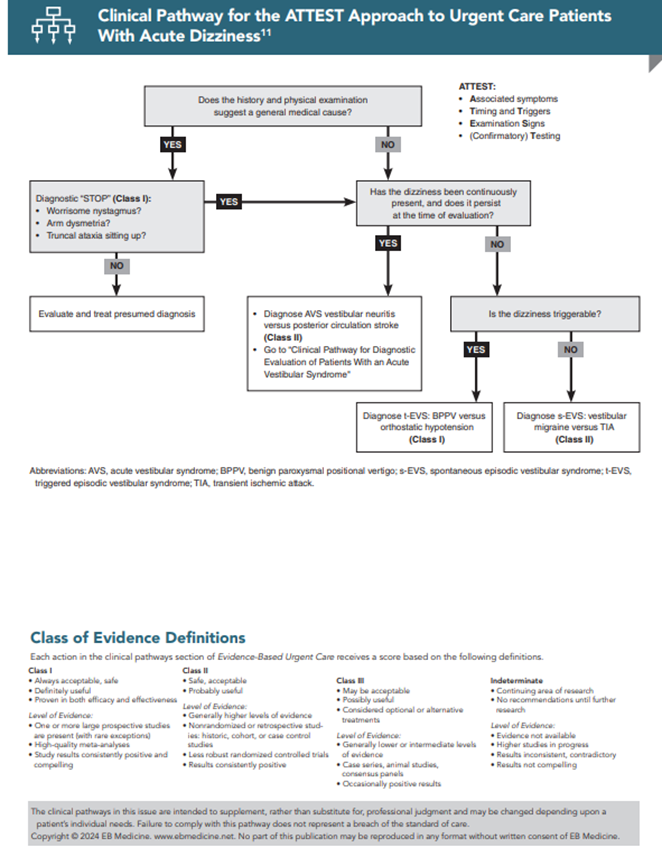
The algorithmic approach to the evaluation of the dizzy patient should begin with ATTEST. (See the Clinical Pathway for the ATTEST Approach to Urgent Care Patients with Acute Dizziness.) The first 3 letters in the ATTEST mnemonic (Associated symptoms, Timing, and Triggers) refer to historical information: “What happened?”, “When?”, “Is the dizziness continuous or intermittent?”, “Are there associated symptoms?” and “What is the broader context?”.
Consider medical causes of the complaint, including:
Each situation suggests a diagnosis or group of diagnoses that would require confirmatory testing. Similarly, the vital signs inform this diagnostic process, i.e., is there fever, tachycardia, hypotension, or hypoxia? Thus, the first diagnostic step in the diagnosis of the patient with acute dizziness is simply to take a history and review the vital signs just as with any other patient. If a general medical diagnosis is likely, I recommend a brief diagnostic “STOP,” which takes less than 1 minute to perform.13,14
To identify patients who might potentially be mimicking a general medical condition, the 3 components of the “STOP” are: (1) a quick test for worrisome nystagmus (see the “Head Impulse–Nystagmus–Test of Skew (HINTS) Testing—Test 1: Nystagmus Testing” section for a detailed description), (2) arm dysmetria, and (3) truncal ataxia. To test for truncal ataxia, simply have the patient sit up on the exam table without grabbing hold of anything for stabilization. If the “STOP” test is reassuring, then proceed with treatment for the presumed condition. If it is worrisome, consider various vestibular or central conditions.
Acute Vestibular Syndrome
If the history does not suggest a general medical condition (or if the “STOP” is worrisome), then the next question to pose is, “Is the dizziness persistently present and still present at the time of evaluation in the UC?” A “yes” answer identifies patients with the AVS, who have the abrupt or rapid onset of dizziness that has lasted hours to days and is still present at the time of examination, even when the patient is lying still. Dizziness may decrease when lying still and worsen with head movement, a common occurrence that does not mean that dizziness has a peripheral cause.
Although the strict neuro-otology definition of AVS includes the presence of nystagmus, some patients who otherwise fulfill the AVS definition (such as many with cerebellar stroke) do not. The presence or absence of nystagmus is a key distinction because it affects how one interprets the HIT.14,1,23,24
Head Impulse–Nystagmus–Test of Skew (HINTS) Testing
Because, by definition, these patients are acutely symptomatic, one can use physical examination to distinguish between central cause (stroke) and peripheral cause (neuritis), referred to as head impulse–nystagmus–test of skew (HINTS) testing. (Note that the HINTS acronym is distinct from the acronym for the head impulse test or HIT.)
An important caveat is that most of the studies that examine the utility of HINTS have been done with neuro-otologists performing the examinations.51,52 One study conducted by stroke neurologists showed that non–subspecialists can be trained to use the HINTS examination effectively.53 Another European study of specially-trained emergency physicians (12 hours of special training using Frenzel lenses to interpret the eye findings) also provided evidence of its effective use in the ED.54,55
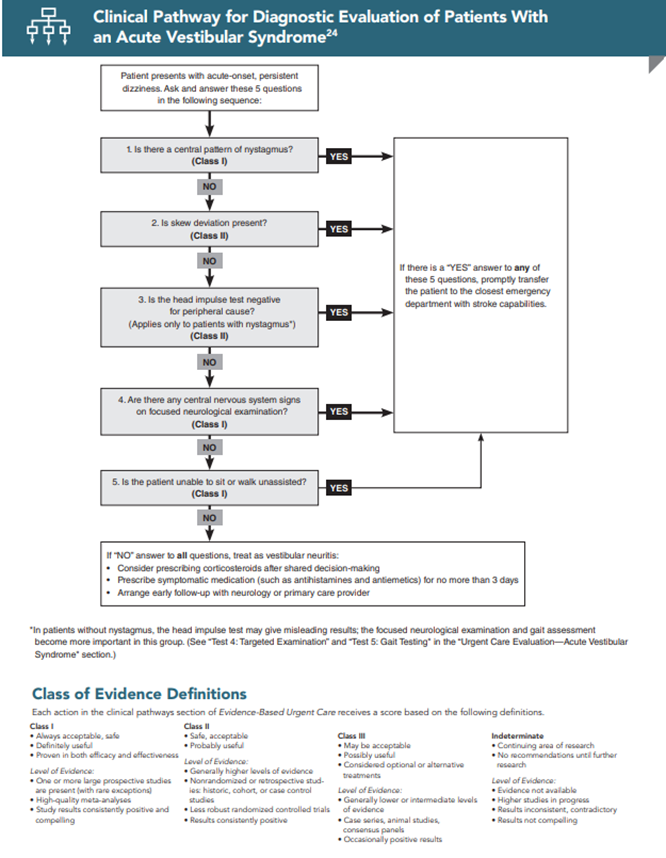
Because HINTS has not been validated in routine practice, 2 additional components must be added to the examination of patients with AVS: a targeted posterior circulation examination and testing of the gait.14,24 When performing the physical examination, these 5 questions should be asked in the following sequence:
None of these tests is 100% sensitive, so if the answer to any one of the questions is “yes,” the patient has a central process, likely stroke, and should be promptly transferred for further workup.14,15,23,24 If the answer to all 5 questions is “no,” then the patient likely has vestibular neuritis and can be safely discharged with outpatient follow-up. The 5 test elements of HINTS testing, in order, are:
Test 1: Nystagmus Testing
The acronym (HINTS) notwithstanding, do not start with the HIT, but rather with nystagmus. There are several reasons for this. First, nystagmus testing is easy for the patient. Second, if there is no nystagmus, then interpretation of the HIT is problematic, since it has been validated only in patients with nystagmus. Third, if there is no nystagmus, it makes the vestibular neuritis and labyrinthitis very unlikely (in patients presenting in the first 2 to 3 days of their illness). Finally, if there is nystagmus that is of a central type, whatever the results of the remainder of the examination, this is a patient who must be assumed to be having a stroke.
To test for nystagmus, first ask the patient to open his eyes and look forward. Observe whether there is any jerk nystagmus, in which the eyes drift in one direction, then snap quickly back. By convention, it is the rapid phase for which the nystagmus is named. If a patient looks forward and his eyes drift to the left, then snap back to the right, he has a right-beating horizontal nystagmus. This is usually very easy to see, especially in the first 2 to 3 days of the patient’s onset of symptoms. Next, ask the patient to follow the examiner’s finger, going 30° to 40° to the right, then to the left. This is called gaze-evoked nystagmus. Also look for vertical or pure torsional nystagmus. In patients with the AVS, nystagmus that is vertical, purely torsional, or that changes direction with the direction of gaze is central.13,15
Test 2: Skew Deviation Testing
Next, check for skew deviation using the alternate cover test, which is also very easy for the patient. Stand in front of the patient, instructing him to focus on your nose. Alternately cover one eye and then the other, multiple times, moving from one eye to the next every second or so. The presence of skew deviation—a small vertical correction in the eye when it is uncovered—indicates a brainstem localization of the patient’s problem. Although uncommon exceptions exist, from the perspective of a clinician, it is safest to assume that skew deviation is always due to a central problem. It is easiest if one focuses on one or the other eye (it does not matter which) because each will display the vertical correction (one going down while the other goes up).
Test 3: Head Impulse Testing
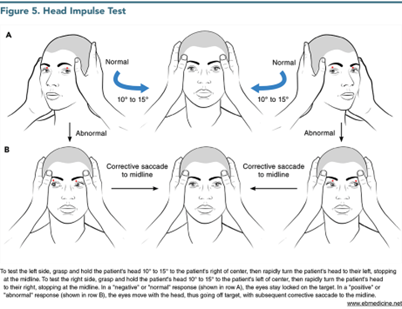
The third component of the examination is the HIT. (See Figure 5.) Again, the patient is instructed to relax his head and neck and to focus on the examiner’s nose. The examiner grasps the patient’s head on both sides and very rapidly snaps it in one direction or the other over a very small arc (only 10°-15°).11,14,15,24 Ideally, hold the head 10° to 15° from the midline and then move it very quickly to the midline. The “normal” or “negative” HIT (when the eyes remain focused on the examiner’s nose) is worrisome for stroke, whereas the “abnormal” or “positive” HIT (when the eyes move with the head and then snap back in one corrective saccade to the examiner’s nose) is reassuring for vestibular neuritis. Therefore, use of the words “normal,” “abnormal,” “negative,” and “positive” to describe the HIT is ambiguous since the “negative” test is worrisome, and the “positive” test is reassuring. It is best to simply state whether a corrective saccade is “absent” or “present.”51,56 Approximately 10% of HITs in which there is a reassuring corrective saccade are false-positives due to strokes,51 usually of the AICA territory or the labyrinth itself.24
Test 4: Targeted Examination
The fourth component of the AVS examination is to perform a targeted examination to detect any central nervous system findings due to posterior circulation ischemia. In addition to a general motor and sensory examination, this examination targets the cranial nerves, cerebellar function, and visual fields. The latter is not in the posterior fossa, but tests the occipital cortex, which is nourished by the posterior cerebral artery, the terminal branches of the basilar artery. This should not require more than a few minutes, but it must be done systematically. Any (new) abnormality indicates a central finding and would therefore be inconsistent with neuritis. For example, anisocoria and ptosis (Horner syndrome) suggest a lateral medullary infarct. Another detail is that the unilateral facial sensory loss in lateral medullary stroke involves pain and temperature, not light touch, which is the usual modality tested by most non–neurologists. It is important to recognize that acute hearing loss, which is traditionally associated with a peripheral process, can also occur with an acute cerebrovascular event involving either the AICA or labyrinthine artery.24
Test 5: Gait Testing
Finally, even if all of the first 4 tests are reassuring, the gait must be tested in patients with AVS. If a patient is unsafe on his feet, he cannot be discharged safely from UC. In addition, the greater the degree of gait abnormality, the more likely it is that the cause of AVS is stroke.57 In a series of 114 patients with AVS (67% with neuritis, 33% with stroke), most patients with neuritis were able to walk independently, whereas most patients with stroke could not.57 In fact, two-thirds of the stroke patients could not even stand up independently. Importantly, all of the 10 patients with AICA stroke (whose HIT can be misleading) had severe gait instability.57
Spontaneous Episodic Vestibular Syndrome
Patients with the s-EVS report one or more episodes of dizziness of variable duration not triggered by head or body-position changes. Because patients with the s-EVS are, by definition, no longer symptomatic and are not triggerable, physical examination is not useful to distinguish the most common diagnoses, which are vestibular migraine and posterior circulation TIA. Diagnosis relies on history and epidemiologic context.13 If a patient with vestibular migraine or TIA was still symptomatic at the time of evaluation, he would present and be evaluated as if he had AVS, just as a patient with an anterior circulation TIA who still had symptoms at the time of presentation would be assumed to be having a stroke.
Specific criteria exist for diagnosis of vestibular migraine.58 There is a strong female predominance for vestibular migraine (5:1).58 Patients with vestibular migraine have multiple episodes of dizziness, and headaches may occur before, during, or after the dizzy episodes.59 When headaches do occur, they are usually (but not always) similar to migraines that occur without the dizziness. The duration of the dizziness is variable and, by definition, can last 5 minutes to 72 hours,60 although rarely the duration is even shorter.58 Because migraine is a central phenomenon, the associated nystagmus can be of a central type.61
Up to half of patients who have posterior circulation TIAs have isolated, transient dizziness.29 Other symptoms include typical posterior circulation symptoms related to the long tracts that pass through the brainstem, cranial nerve dysfunction, or visual field cuts due to posterior cerebral artery ischemia of the visual cortex. Contrary to conventional wisdom, short-term stroke risk may be higher with posterior circulation TIA than with anterior circulation TIA.28,62
Recognizing that none of these elements can be used in a binary, yes/no fashion, factors that suggest vestibular migraine over TIA include younger age, more frequent attacks over a longer period of time, other migraine-related symptoms (such as headache, phonophobia, photophobia), and absence of traditional vascular risk factors.
Patients with Ménière disease (which was relatively uncommon in an ED series of dizzy patients) also present with s-EVS and will usually have ringing or buzzing in the ear and, over time, progressive hearing loss.63 Treatment is symptomatic, and patients should be referred to an ENT specialist.
Triggered Episodic Vestibular Syndrome
The physical examination is very helpful in patients with a t-EVS and will often establish a specific diagnosis. Although the utility of orthostatic vital signs has been traditionally downplayed, a patient with dizziness when standing up who develops symptoms and orthostatic vital sign changes on standing up is highly likely to have orthostatic hypotension as a cause of the dizziness, and the evaluation is directed at finding the underlying cause.
BPPV should be suspected in patients with very brief episodes of dizziness, generally lasting less than a minute. Brief episodes of dizziness that wake a patient up from sleep are nearly always BPPV.64-67 One study showed a positive likelihood ratio of 60 for a BPPV diagnosis if dizziness occurred with lying down or turning in bed.66
In patients with suspected BPPV, bedside testing can confidently establish the diagnosis. The most commonly affected canal is the posterior canal (pc-BPPV) which is usually tested by the Dix-Hallpike maneuver. If this test is negative on both sides, then the horizontal canal (hc-BPPV) is tested by the supine head roll test. In pc-BPPV, the nystagmus is typically up-beating and torsional, and in hc-BPPV, it is horizontal and direction-changing. This illustrates how the interpretation of nystagmus differs from AVS (where torsional or direction-changing = worrisome diagnosis) from t-EVS (where torsional for pc-BPPV and horizontal direction-changing for hc-BPPV = benign diagnosis).
Occasionally, BPPV patients have no nystagmus.68-70 Possible causes are a small number of otoliths in the canal, use of vestibular suppressants at the time of diagnosis, or small-amplitude nystagmus that the examiner is not perceiving due to visual fixation by the patient.
Some patients with hc-BPPV will have spontaneous (or more persistent) nystagmus that is normally not seen with BPPV.71,72 This occurs because, depending on the orientation of the patient’s head, otoliths in the horizontal canal may be moving in a patient sitting up and looking forward.
Finally, very rarely, patients with CPPV caused by structural lesions adjacent to the fourth ventricle (usually a tumor, multiple sclerosis plaque, or small brainstem stroke) will exhibit nystagmus or other features that are atypical for BPPV.34,73 These patients will often have some symptoms (such as headache) that patients with BPPV never have, or they do not respond as expected to a repositioning maneuver. They may have physical findings that localize to the brainstem or cerebellum that patients with BPPV do not have, or they may exhibit nystagmus in the absence of movement or dizziness.
Risk Management Pitfalls for Dizziness in the Urgent Care Setting
Excerpted from: Toscano J. The timing-and-triggers approach to the Urgent Care patient with acute dizziness. Evidence-Based Urgent Care. 2024 February;3(2):1-27. [Content was adapted from: Edlow JA. The timing-and-triggers approach to the patient with acute dizziness. Emerg Med Pract. 2019;21(12):1-24. Used with permission of EB Medicine.] Reprinted with permission of EB Medicine.
References